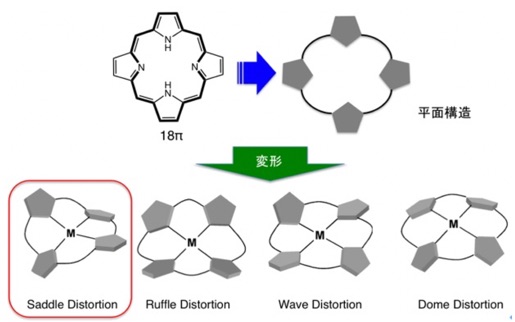
Porphyrin molecules and structural distortion

Porphyrins are 18π-electron heteroaromatic compounds that exist in vivo as iron complexes in the active centers of hemoglobin and myoglobin. Porphyrins are characterized by their strong absorption in the visible region, their redox activity, and their ability to form metal complexes, serving as dianionic macrocyclic ligands for various metal ions. Conventional porphyrin molecules exhibit a planar structure due to their extended π-conjugation system.
Our laboratory is conducting research on dodecaphenylporphyrin (H2DPP), which has 12 phenyl groups introduced to the periphery, and its derivatives. DPP derivatives have a distorted saddle-shaped structure due to steric repulsion between the phenyl groups. We are working to develop new functions for porphyrins by taking advantage of the following characteristics derived from their saddle-distorted structure.
(1) When a metal complex is formed, the Lewis acidity of the central metal is improved compared to that of a normal planar porphyrin complex, allowing stronger axial coordination.
(2) The interaction between the lone pairs of pyrroles and the center metal is weakened, lowering the oxidation potential of the porphyrin ring. The electron-donating property in electron transfer reactions is improved.
(3) Diprotonated porphyrin dication is easily and stably formed, and the redox potential is elevated. It functions as an electron acceptor in electron transfer.
(4) Stable supramolecular structures can be constructed by utilizing the strong hydrogen bonding with anionic species in the diprotonated form.