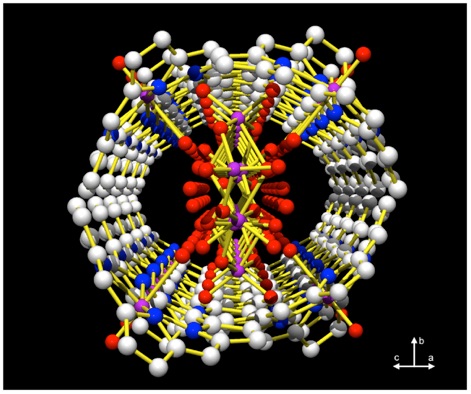
A porphyrin nanotube is formed via self-assembly through π-π interactions between peripheral phenyl groups of a saddle-distorted molybdenum(V)- dodecaphenylporphyrin (DPP) complex, concomitant with the inclusion of tetranuclear molybdenum(VI)-oxo clusters with unique structures in their closed π spaces through hydrogen bonding (Angew. Chem. Int. Ed. 2004, 43, 1825; Chem. Mater. 2007, 19, 51 (Cover Picture)). This is the first example that the crystal structure of a nanosized tubular porphyrin supramolecule, whose crystal structure has been determined.


Crystal structure of porphyrin nanotubes Based on Self-Assembly of Mo(V)−Dodecaphenylporphyrin Complexes and Inclusion of tetranuclear Mo-oxo clusters
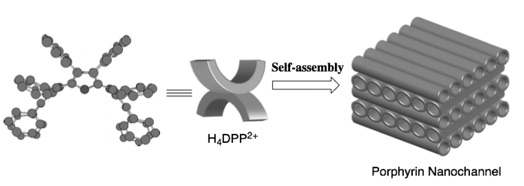
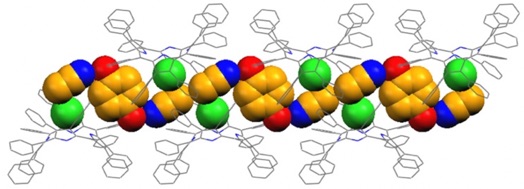
The hydrochloride salt of H2DPP, [H4DPP]Cl2, is crystallized from chloroform-acetonitrile to form a supramolecular structure named "porphyrin nanochannel (PNC)." In this supramolecular structure, water molecules, chloroform and acetonitrile molecules are encapsulated in the nano-sized guest inclusion site (PNC-water). Furthermore, when an electron-donating molecule such as hydroquinone derivatives is coexisted, a "PNC-guest" is formed by selectively incorporating the guest molecules into the inclusion site. In contrast, in the presence of quinones, which are electron-accepting molecules, PNC-water is formed without inclusion of quinones. That is, PNC can recognize the electronic properties of guest molecules and encapsulates them .(Chem. Commun. 2005, 716; Chem. Eur. J. 2007, 13, 8212) . Furthermore, when visible light is irradiated onto PNC encapsulating an electron-donating molecule, photoinduced electron transfer from the electron-donating guest molecule to H4DPP2+ occurs, and the PNC exhibits photoconductivity .(Chem. Mater. 2008, 20, 7492; J. Mater. Chem. 2008, 18, 1427) .
Crystal structure of a porphyrin nanochannel derived from self-assembly of H4DPP2+Cl2 including hydroquinone
Insufficient overlap between the lone pair orbitals of saddle-distorted porphyrin and the metal d-orbitals results in insufficient electron donation to the metal center, increasing the Lewis acidity of the central metal more than that of metal centers bound to planar porphyrins. This strengthens the axial coordination at the metal center and allows the formation of integrated structures that are not possible with planar porphyrin complexes. As an example, a “porphyrin hamburger” as a discrete complex is formed by direct coordination of terminal oxo groups of heteropolyoxometalates (POMs), whose size, shape and charge can be clearly controlled, to the Mo(V) center of [Mo(DPP)(O)(H2O)]+ via a ligand exchange reaction. (Chem. Commun. 2007, 3997 (Selected as a Cover Picture); Highlighted in Chem. Sci. 2007, 4, C58). Porphyrin hamburger retains its structure stably even in solutions such as benzonitrile and exhibits reversible multistep redox behavior (Inorg. Chem. 2010, 49, 11190).
Crystal structure of Porphyrin Hamburger
[{Mo(DPP)(O)}2(H2SiW12O40)]
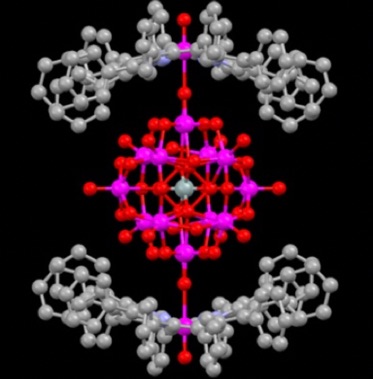
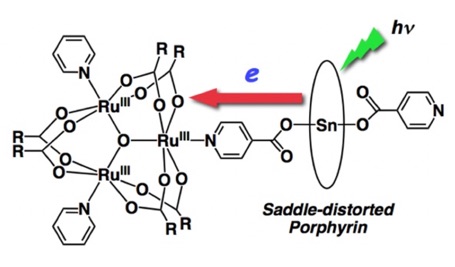
SnIV-DPP complexes are effective for developing novel photofunctional molecules and materials by utilizing their strong axial coordination and electron-donating ability. We have constructed conglomerates by linking saddle-distorted SnIV(DPP) complexes to trinuclear RuIII-oxo clusters having 4-pyridinecarboxylate through the coordination of the carboxylate group to the Sn(IV) center. Within the complex, nonadiabatic photoinduced electron transfer occurs from the SnIV-DPP moiety to the ruthenium cluster. Interestingly, in this complex, the electron transfer from the Sn(DPP) moiety to the Ru trinuclear cluster and the back electron transfer both fall in the Marcus inverted region because the reorganization energy of electron transfer is small (Chem.–Eur. J. 2010, 16, 3646; figure below).