Development of new functions based on protonation

In planar porphyrins, protons of pyrrole are delocalized, and an excess of Brφnsted acid is required for the protonation of the pyrroles. In contrast, in saddle-distorted porphyrins, the lone electron pair of pyrrole is easily accessible, allowing quantitative protonation even with weak acids such as carboxylic acids. Protonated H2DPP can not only form hydrogen bonding with the conjugate base to form supramolecules but also function as an electron acceptor in electron transfer because its redox potential is significantly lowered by protonation (J. Am. Chem. Soc. 2009, 131, 577, ; Coord. Chem. Rev. 2012, 256, 2488).
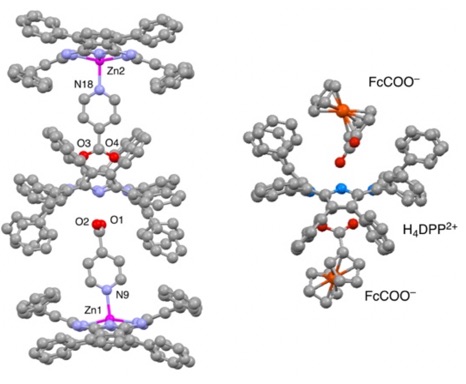
When an electron donor with a carboxyl group is reacted with H2DPP, a donor-acceptor-donor supramolecule is formed, in which the electron donor is hydrogen-bonded above and below H4DPP2+. The DOSY NMR spectra of the supramolecular assembly in CDCl3 revealed that the supramolecular assembly composed of saddle-distorted phthalocyanine on the left in the figure below, the Zn(II)–phthalocyanine complex coordinated by 4-pyridincarboxylate as a ligand, and the hydrogen bonding between the carboxylate group and H4DPP2+ is maintained in solution. When this supramolecular assembly is photoexcited, photoinduced intrasupramolecular electron transfer from the phthalocyanine complexes to H4DPP2+ occurs to generate an electron-transfer state with the lifetime of 667 ps (Angew. Chem. Int. 2008, 47, 6712).
This phenomenon has been generalized. [H4DPP](Donor-COO)2, supramolecular assemblies formed by hydrogen bonding between H4DPP2+ and electron donors (Donor; D) with a carboxylate group, was prepared. As an example, the supramolecule containing ferrocene carboxylate derivatives (FcCOO–) is shown on the right in the figure. Photoinduced electron transfer from the donor moieties to H4DPP2+ in these supramolecules is non-adiabatic, and the rate constant of the electron transfer shows a slightly smaller D-A distance dependence (β = 0.64 Å–1) due to the strong hydrogen bonding (JACS 2010, 132, 10155).
We have found that H2DPP can form a monoprotonated porphyrin (H3DPP+) by controlling acid strength, and succeeded in the thermodynamic control of its formation. Additionally, we also explored photoinduced intermolecular electron transfer from electron donors to the monoprotonated H2DPP (Chem. Commun. 2009, 4994; Chem. Eur. J. 2017, 23, 4669).
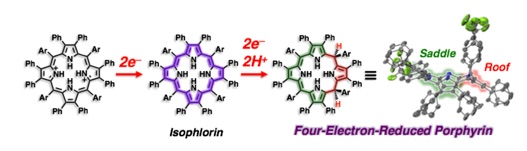
A stable non-aromatic 20π isophlorin derivative is formed from a diprotonated H2DPP derivative having an electron-withdrawing groups through the two-electron oxidation. Furthermore, as the isophlorin derivative undergoes further two-electron reduction in DMSO, a novel four-electron reduced porphyrinoid was obtained to determine its crystal structure. This four-electron reduced species is oxidized by quinone (DDQ) and quantitatively returns to the original porphyrin diprotonated form. (Angew. Chem. Int. Ed. 2018, 57, 1973)
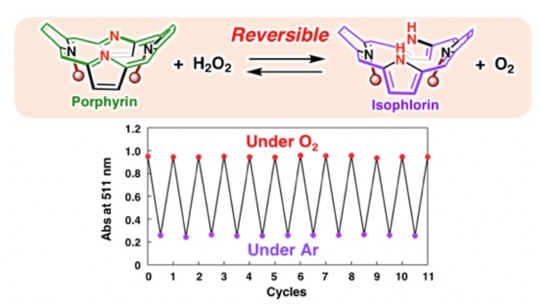
The DPP derivative (Me2DPP) with methyl groups introduced to nitrogen atoms in its two pyrroles showed unprecedented redox reactivities. Me2DPP reacts with hydrogen peroxide (H2O2) to be reduced to an isophlorin derivative; this isophlorin derivative is oxidized back to Me2DPP by reacting with oxygen (O2). In other words, based on the reversible redox process between Me2DPP and H2Me2DPP, the interconversion between oxygen(O2) and hydrogen peroxide(H2O2) has been achieved.(J. Am. Chem. Soc. 2019, 141, 5987)
Multi-Electron Reduced Porphyrinoids Formed by Chemical Reduction of H4DPP2+ Derivatives