Catalysis by protonated porphyrins

Based on the electron-accepting properties of H4DPP2+, we have been developing photocatalytic reactions. First, we have developed an oxidation reaction of organic substrates in aqueous solution, using a water-soluble H4DPP2+ derivative as a photosensitizer and a ruthenium-substituted Keggin-type heteropolyoxometalate (RuPOM) as an oxidation catalyst. In this catalytic system, a 2:1 supramolecular assembly is formed between the water-soluble H4DPP2+ derivative and RuPOM, and the oxidant S2O82– also associates with the water-soluble H4DPP2+ derivative. Therefore, after photoinduced electron transfer from RuPOM to the excited singlet state of the water-soluble H4DPP2+ derivative, electron transfer occurs from the reduced porphyrin to S2O82– that is associated with the porphyrin, generating a Ru(V)-oxo complex as an oxidative active species and causing the substrate oxidation reaction to proceed.
(Green Chem. 2018, 20, 1975)
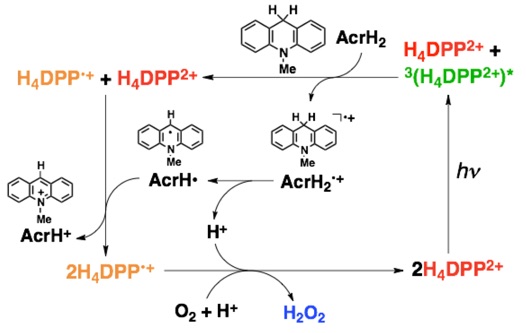
H4DPP2+ functions as a self-photosensitizing catalyst for the highly selective 2-electron/2-proton oxygen reduction reaction to produce hydrogen peroxide (H2O2). First, using 10-methyl-9,10-dihydroacridine (AcrH2) as a reductant, under visible light irradiation in the presence of H4DPP2+ and protons, oxygen (O2) is selectively reduced to hydrogen peroxide (H2O2). The quantum yield of this reaction is determined to be 12%, and the turnover frequency (TOF) of the catalyst is determined to be 500 h⁻¹. In this photocatalytic reaction, the excited triplet state of H4DPP2+ is at a lower energy level than that of a typical planar porphyrin, making it important that the photoinduced electron transfer from the electron donor proceeds preferentially over energy transfer to oxygen (O2). (Chem. Commun. 2019, 55, 4925)
Photocatalytic substrate oxidation reaction in aqueous solution using H4DPP2+ as a photosensitizer
Generation of hydrogen peroxide(H2O2) by oxygen reduction under visible light irradiation using H4DPP2+ as a photocatalyst
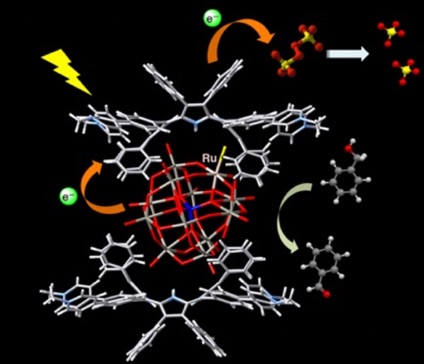
On the other hand, a photo-hydrogen generation system was constructed using H4DPP2+ as a photocatalyst, 10-methyl-9,10-dihydroacridine (AcrH2) as a two-electron donor, and Pt nanoparticles (PtNPs) as a catalyst. In this catalytic system, the hydrogen evolution reaction proceeds with a quantum yield of 17% under near-infrared light irradiation at 710 nm. It was found that the rate-determining step of this photo-hydrogen generation is the photoinduced electron transfer from AcrH2 to the excited triplet state of H4DPP2+. (ACS Appl. Energy Mater. 2020, 3,3193)
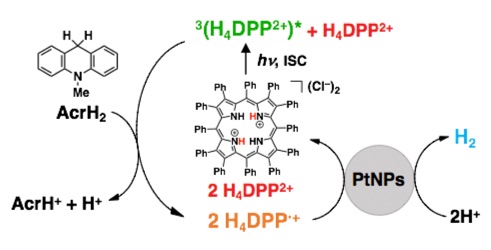
A highly efficient NIR light-driven H2 evolution system using H4DPP2+ as a photocatalyst
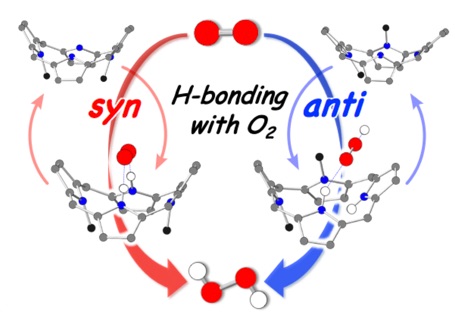
Furthermore, the N-methylated DPP derivative (Me2DPP) catalyzes the generation of hydrogen peroxide(H2O2) via two-electron reduction of oxygen(O2), proceeding through an intermediate of isophlorin derivative formed by thermal electron transfer in the presence of proton and electron sources. In this catalytic reaction, the reaction mechanism dramatically changes depending on the positions where the methyl groups are introduced. In the syn-Me2Iph, the intermediate of isophlorin derivative with the two pyrrole protons facing the same direction forms two-point hydrogen bonds with the oxygen molecule, promoting the oxygen reduction reaction. In contrast, in the anti-Me2Iph, the pyrrole proton forms a hydrogen bond with oxygen at one point, and the reaction is accelerated by external protons. Kinetic analysis of the oxygen reduction reaction revealed that the rate-determining steps differ in each case.(Chem. Eur. J. 2020, 26, 10480 (Very Important Paper))