Nakamura Laboratory, Research Outline
Natural receptor proteins can bind substrates selectively at the pocket surrounded by multiple amino acid residues. Relatively weak intermolecular interactions, such as hydrogen bonds, are synergistically exerted during the recognition events. Meanwhile, it was difficult for synthetic receptors to achieve precise molecular binding by arranging various interaction moieties in an unsymmetrical manner.
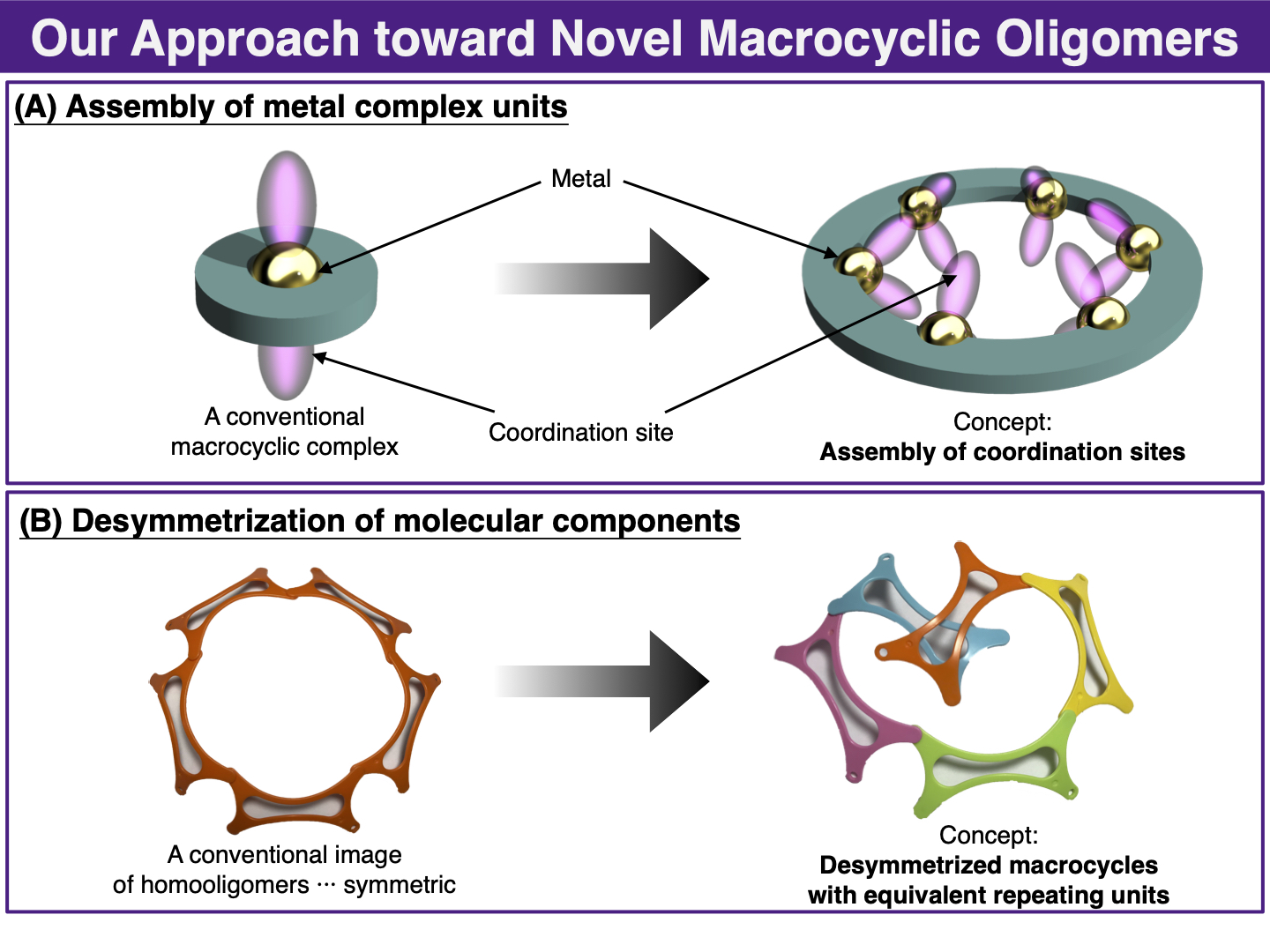
Our laboratory develops novel macrocyclic receptors with precise recognition properties based on two concepts, that is, (A) assembly of metal coordination sites, and (B) desymmetrization of the molecular components
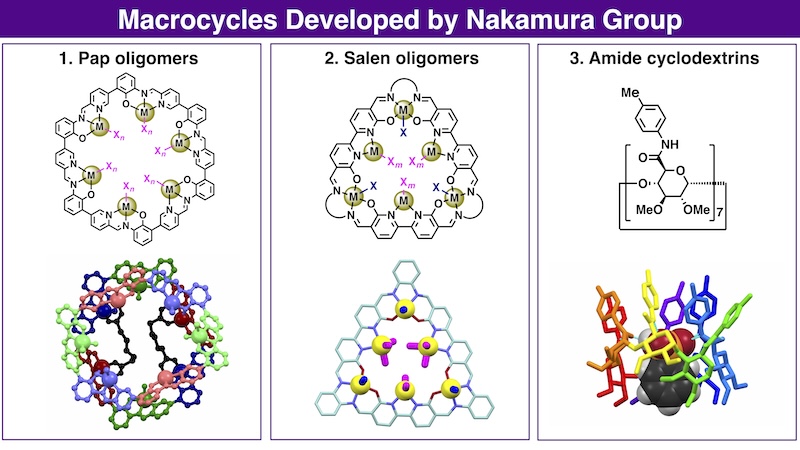
- Pap and sap oligomers: Receptors that capture molecules via multipoint coordination
(a) Nature Commun., 8, 129 (2017). (Paper)
(b) Chem. Commun., 55, 2421-2424 (2019). (Paper) (Inside Front Cover)
(c) Eur. J. Inorg. Chem., 308-313 (2021). (Paper) (Cover Feature)
(d) Inorg. Chem., 62, 12886-12894 (2023). (Paper) (Supplementary Cover)
(e) Chem. Commun., 60, 1281-1284 (2024). (Paper)
(f) Chem. Eur. J., 31, e202500424 (2025). (Paper)
(g) Dalton Trans., 54, 6304-6307 (2025). (Paper)
(h) Angew. Chem. Int. Ed., 64, e202505734 (2025). (Paper) - Salen oligomers: Spatial arrangement of metals and their coordination sites
(a) J. Am. Chem. Soc., 141, 6462-6467 (2019). (Paper)
(b) Inorg. Chem., 58, 7863-7872 (2019). (Paper) (Supplementary Cover)
(c) Inorg. Chem., 63, 12697–12702 (2024). (Paper)
(d) Chem. Commun., 61, 921-924 (2025). (Paper)
- Amide and carboxy cyclodextrins: Recognition utilizing multipoint hydrogen bonds
(a) Chem. Commun., 55, 3872-3875 (2019). (Paper) (Inside Front Cover)
(b) Chem. Lett., 49, 493-496 (2020). (Paper)
(c) Angew. Chem. Int. Ed., 60, 3080-3086 (2021). (Paper)
(d) Chem. Sci., 16, 171-181 (2025). (Paper)
(e) J. Org. Chem., 90, 3603-3609 (2025). (Paper)
(f) J. Org. Chem., 90, 11452-11459 (2025). (Paper)
Recent Reviews and Books
- “Supramolecular complexes that capture molecules by multi-point coordination bonds
”
Bull. Jpn. Soc. Coord. Chem., 70, 39-42 (2017). - “Development of Artificial Receptors Based on Assembly of Metal Complex Units and Desymmetrization of Molecular Components”
Chem. Lett., 50, 1822–1830 (2021). (Award Highlight Review) (Paper) (Inside Cover)


